REAL-TIME PRESSURE AND TEMPERATURE MONITORING OF PHOTO-IMMUNOTHERAPY USING A FIBER OPTIC SENSOR
PRINCIPAL INVESTIGATORS:
Hisataka Kobayashi, Center for Cancer Research, National Cancer Institute
Peter Choyke, Center for Cancer Research, National Cancer Institute
Miao Yu, Department of Mechanical Engineering, University of Maryland
PH.D. STUDENT:
Zhijian Zhang, Mechanical Engineering, University of Maryland
Photo-immunotherapy (PIT) is a newly established highly target-specific cancer therapy based on an armed antibody conjugate that induces rapid cellular necrosis after exposure to near infrared light. The conjugate consists of a hydrophilic photosensitizer phthalocyanine dye, IR700, which is covalently bound to a humanized monoclonal antibody (mAb). A variety of approved mAb, targeting cancer-specific cell-surface molecules, have been successfully used for PIT. When exposed to NIR light, the conjugate induces highly selective rapid, necrotic, cancer cell death both in vitro and in vivo [1]. Following PIT, massive necrotic changes occurs only in cancer cells but not in normal tissue so that tumor vasculature is preserved [2]. As a result, there is enhanced delivery of nano-sized drugs which is up to 25-fold more than untreated tumors. This phenomenon has been termed “super-enhanced permeability and retention effects (SUPR) to differentiate it from enhanced permeability and retention (EPR) [3]. To better understand this phenomenon, during PIT-therapy it is important to measure physiological parameters, such as interstitial pressure, and temperature. High interstitial pressure is believed to prevent efficient drug delivery[4]. We hypothesize that PIT reduces tumor interstitial pressure thus enabling the SUPR effect. Therefore, we propose a real-time miniature optical pressure sensor that will report changes occurring during PIT. Conventional piezoelectric pressure transducers are not ideal for this application due to their large size relative to the tumor. Here, we propose to develop a miniature, polymer based fiber optic pressure and temperature sensor for use with PIT. A polymer based fiber optic sensor (O.D. of 150 µm) has been developed by Prof. Yu’s group at UMD, which has been demonstrated to have the capability of simultaneous temperature/pressure measurements with high sensitivity [5][6]. With the proposed sensor, pressure and temperature of tumor can be monitored with high accuracy but minimal invasiveness during the PIT which is critical to understanding the timing of pressure/temperature changes in situ.
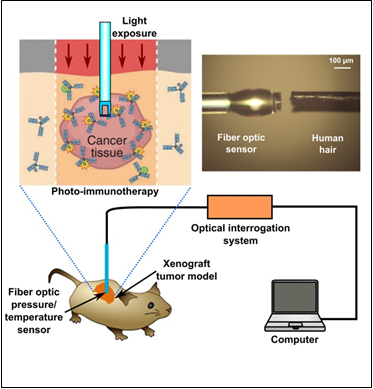
PIT is a molecular targeting cancer therapy with minimum side effects. PIT has been demonstrated to effectively treat cancer cells without damaging surrounding normal tissue. To better understand and optimize the effectiveness of this therapy, it is important to measure and study the change of physiological parameters (e.g. interstitial pressure, temperature, etc). This project will develop a miniature multifunctional fiber optic sensor, which provides a real-time monitoring tool to study and monitor PIT therapeutic effect in vivo.
MODELING PATHWAYS TO THERAPEUTIC TARGETING OF HIGH-GRADE SEROUS OVARIAN CANCER AND DRUG RESISTANCE
PRINCIPAL INVESTIGATORS:
Jung-Min Lee, National Cancer Institute
Doron Levy, Department of Mathematics, University of Maryland
PH.D. STUDENT:
Dana-Adriana Botesteanu, Applied Mathematics, Statistics, and Scientific Computation, University of Maryland
The goal of this project is to study the evolution of high-grade serous cancer in the face of treatment and drug resistance. Specifically, we seek to describe a putative mechanism of high-grade serous ovarian cancer recurrence in the presence of treatment: the emergence of revertant secondary mutations restoring BRCA1/2 function in a number of platinum- resistant carcinomas. We will use mathematical tools (ordinary, partial and delay differential equations, and optimal control) to develop a mathematical framework starting from (early) microscopic molecular events to the (subsequent) macroscopic evolution of high-grade serous ovarian cancer prior to therapy under different pathological scenarios.
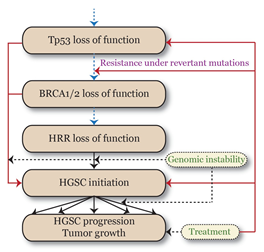
The underlying structure of the proposed mathematical model. Highlighted is the temporal sequence of events associated with initiation and progression of high-grade serous cancer from premalignant to invasive cancer. Dashed blue arrows represent slow-occurring events, solid black arrows rate-limiting events, and dashed black arrows represent fast-occurring events induced by genomic instability. Solid red arrows show high-grade serous cancer response induced by the treatment.
DISSECTING THE GENE REGULATORY FUNCTION OF MYC WITH SINGLE-MOLECULE ANALYSIS
PRINCIPAL INVESTIGATORS:
Daniel Larson, National Cancer Institute
David Levens, National Cancer Institute
Michelle Girvan, Department of Physics, University of Maryland
PH.D. STUDENT:
Simona Patange, Biophysics, University of Maryland
Gene expression refers to the sum of processes that enable cells to control their complement of RNA, and the study of gene expression has been spurred by post-genomic biochemical techniques such as microarrays and RNA sequencing. In recent years, through parallel advances in microscopy, fluorescent probe development, and computational modeling, it has become possible to describe gene expression in a fundamentally different way: one can now directly observe single molecules of RNA in living and fixed cells using the fluorescence microscope. The view that has emerged from these studies is that stochastic effects- 'noise' in gene expression - influences phenotype selection and disease progression. The MYC transcription factor, which is upregulated in >50% of human cancers, has recently been shown to regulate a large fraction of genes in the human genome by an unknown mechanism. We propose to use single-molecule approaches in intact cells to determine how MYC alters stochastic gene regulation.
PREVENTING SEVERE HEARING LOSS AND DEAFNESS IN CHEMOTHERAPY PATIENTS
PRINCIPAL INVESTIGATORS:
Andrea Apolo, Center for Cancer Research, National Cancer Institute
Ben Shapiro, Fischell Department of Bioengineering, University of Maryland
PH.D. STUDENT:
Bharath Ramaswamy, Bioengineering, University of Maryland
Platinum based chemotherapy drugs, such as Cisplatin, are known to cause severe hearing loss, tinnitus and vertigo in patients. In addition to being a major quality of life issue, unacceptable hearing loss is a major reason to prevent or cut short cisplatin regimens in patients.
Our group has developed a method to locally deliver otoprotectants into the cochlea, thus providing the potential to save hearing without interfering with the systemic action of cisplatin chemotherapy. In this UMD‐NCI research, we will test if our treatment can save hearing in a clinically relevant animal model of cisplatin-induced hearing loss.
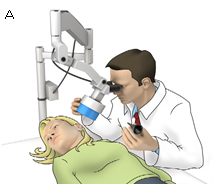
Schematic of the treatment as it would be used in a cancer patient. The treatment employs magnetic forces to direct therapy into the cochlea and is minimally-invasive.
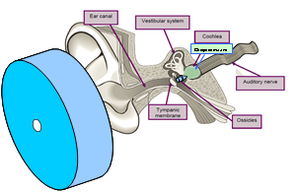
Ear diagram shows device placement, ear anatomy including the location of the cochlea, and the delivered otoprotectant (light green).