PREDICTING PATIENTS RESPONSE TO PREOPERATIVE CHEMO-RADIOTHERAPY FOR RECTAL CANCER
PRINCIPAL INVESTIGATORS:
Thomas Ried, National Cancer Institute
Eytan Ruppin, Department of Computer Science and Center for Bioinformatics & Computational Biology (CBCB), University of Maryland
PH.D. STUDENT:
Noam Auslander, Computer Science, University of Maryland
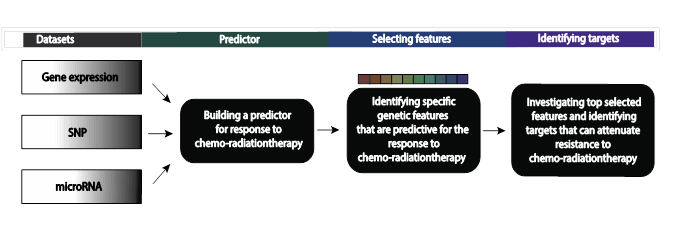
Rectal carcinomas account for approximately 20% of all colorectal cancers. Patients with stage II and III rectal carcinoma are treated with chemo-radiotherapy (CRT) before surgery to reduce the rate of local recurrences. However, not all patients respond equally well to CRT, with response ranging from complete response, i.e., no tumor cells left, to primary resistance. Clearly, accurately predicting the response to CRT before treatment commences would be immensely useful: patients whose tumors are resistant could either be treated with other regimens, or by surgery alone. Alternatively, patients with a predicted complete response and having other comorbidities might be spared surgery. Our goal is to build a predictor that will predict response to CRT based on the molecular data characterizing the tumor of each individual patient, spanning its gene expression, single nucleotide polymorphism and microRNA data. This analysis will also reveal the set of discriminating genes and accompanying molecular features that play a key role in modulating the response to CRT in rectal cancer. This will advance our understanding of the biological mechanisms behind resistance to chemo-radiotherapy in rectal cancer and may lead to new targets whose inhibition may mitigate this resistance.
MECHANICAL REGULATION OF NUCLEAR AND TRANSCRIPTION FACTOR DYNAMICS
PRINCIPAL INVESTIGATORS:
Gordon Hager, National Cancer Institute
Arpita Upadhyaya, Department of Physics, University of Maryland
PH.D. STUDENT:
David A. Garcia Grisales, Physics, University of Maryland
Cells can sense and respond to the physical properties of their surroundings such as stiffness, geometry and topography. This research aims to understand how changes in the mechanical environment of cells modulate the properties of their gene expression patterns. Combining the expertise in the Hager lab at the NCI and the Upadhyaya lab at UMD, researchers will examine how the dynamics of transcription factors in the nucleus is modulated by changing the shape of the cells and the stiffness of the matrix they are grown on. This research has the potential to uncover fundamental mechanisms underlying mechanosensing in cancer cells.
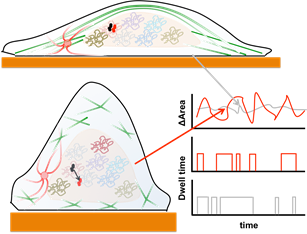
Schematic showing how changes in cell shape result in altered actin cytoskeleton (elongated shape and stress fibers v/s isotropic shape with small actin networks. The elongated cell is associated with smaller nuclear shape fluctuations (gray) v/s larger fluctuations (red) and altered statistics of transcription factor diffusion and occupancy.
RATIONAL DESIGN OF SELF-ASSEMBLED NUCLEIC ACID ADJUVANTS TO PROGRAM TUMOR IMMUNITY
PRINCIPAL INVESTIGATORS:
Bruce Shapiro, Center for Cancer Research, National Cancer Institute
Joost Oppenheim, Center for Cancer Research, National Cancer Institute
Christopher Jewell, Fischell Department of Bioengineering, University of Maryland
PH.D. STUDENT:
Neil Dold, Bioengineering, Unviersity of Maryland
Therapeutic cancer vaccines that arm immune cells against antigens over-expressed on cancer cells have the potential to transform cancer therapy. However, these vaccines have been hindered by1) inefficient expansion of T cells against tumor antigens, 2) an inability to maintain anti-tumor response in the presence of the immune-suppressive tumor microenvironment, and 3) poor generation of tumor-specific T cell memory to prevent relapse. More rational adjuvant design could address these challenges by activating more robust sets of pro-immune pathways that drive potent and durable expansion of tumor-specific T cells. In the proposed research (see figure below), potent RNA-based adjuvants will be designed and loaded in controlled release polymer particles (Aim 1), then the ability of the particles to generate and polarize T cell function against tumor antigens will be tested in vitro (Aim 2) and in mouse models of liver cancer and thymoma (Aim 3). These aims will be carried out by a talented graduate student, Mr. Neil Dold, with the support of a synergistic team that merges the biomaterials and bioengineering skills of Dr. Chris Jewell (UMD), RNA biology and computational knowledge of Dr. Bruce Shapiro (NCI), and the translational cancer expertise of Dr. Joost Oppenheim (NCI).
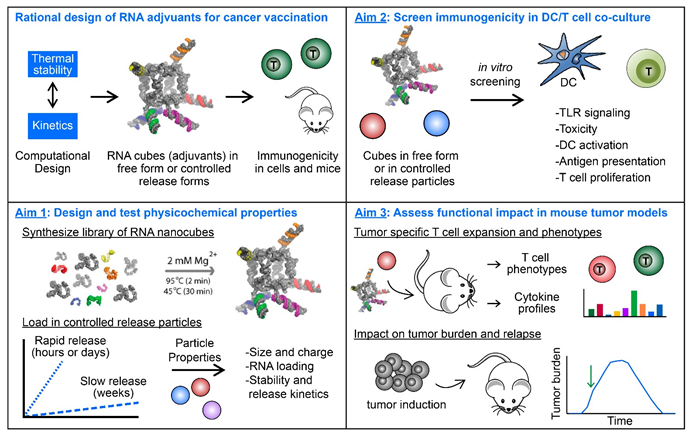
Approach to design and test controlled release of RNA adjuvants for enhanced cancer vaccination
DISSECTING TUMOR INFILTRATING LYMPHOCYTE DIVERSITY BY SINGLE-CELL TRANSCRIPTOME ANALYSIS
PRINCIPAL INVESTIGATORS:
Remy Bosselut, Center for Cancer Research, National Cancer Institute
Sridhar Hannenhalli, Department of Cell Biology and Molecular Genetics, University of Maryland
PH.D. STUDENT:
Assaf Magen, Computer Science, University of Maryland
Cancers generate complex immune responses, aimed at eliminating tumor cells or inhibiting their growth but typically neutralized through multiple evasion strategies. Tumor-infiltrating T lymphocytes have emerged over the past decade as major controllers of tumor growth or rejection, although their activity can be highjacked by cancer cells to support proliferation or metastasis. Because tumor infiltrating lymphocytes are highly diverse and can exert both negative and paradoxically positive effects on tumor progression, dissecting their heterogeneity is critical to understand their impact on tumor progression, harness their power to destroy tumor cells and therefore to improve or design cancer immunotherapy strategies. Here, we propose to address this challenge through single-cell analysis of RNA expression by deep sequencing (RNA-seq), using a novel single cell RNA-seq protocol and designing state-of-the-art bioinformatics tools.

The cartoon depicts tumor-infiltrating lymphocytes (TIL, filled circles), a highly diverse population including cells opposing tumor growth and cells ‘diverted’ to support proliferation of cancer cells (blue shapes) or metastasis. Because tumor infiltrating lymphocytes. We propose to dissect TIL heterogeneity by single-cell RNA-seq to understand and predict their impact on tumor progression and improve cancer immunotherapy strategies.
CONTACT GUIDANCE DURING BREAST CANCER CELL METASTASIS
PRINCIPAL INVESTIGATORS:
Carole Parent, National Cancer Institute
Stanley Lipkowitz, National Cancer Institute
Wolfgang Losert, Department of Physics and Institute for Physical Science and Technology, University of Maryland
John Fourkas, Department of Chemistry and Biochemistry, University of Maryland
PH.D. STUDENT:
Song Chen, Biophysics, University of Maryland
Tumor metastasis is the primary cause of cancer-related deaths. The important question we aim to address is whether nanotopographies can trigger changes in cytoskeletal dynamics and induce cancer cells to exhibit distinct dissertation patterns, and what are the mechanisms underlying this topographic regulation. We hypothesize that contact guidance regulates cancer dissemination by fostering directional cell migration and by changing collective cell behavior toward more invasive phenotypes. We will study nanotopography-induced contact guidance using a breast cancer cell progression model and monitor cytoskeletal dynamics by live cell fluorescent imaging and quantitative image analysis.
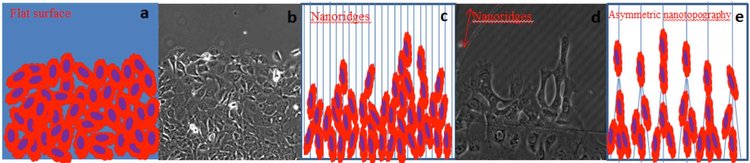
(a), (c) and (e) are cartoons depicting cancer cells migrating collectively on different nanotopographic surfaces. (b) and (d) are images of MCF10A cells migrating on a flat surface and nanoridges, respectively. We hypothesize that nanotopographies regulate collective cell migration toward more invasive phenotypes.