HIGH-RESOLUTION ATOMISTIC SIMULATIONS AND EXPERIMENTAL CHARACTERIZATIONS OF CENP-A/H3 HYBRID NUCLEOSOMES
PRINCIPAL INVESTIGATORS:
Yamini Dalal, National Cancer Institute
Garegin Papoian, Department of Chemistry and Biochemistry, University of Maryland
PH.D. STUDENT:
Mary Pitman, Chemistry, University of Maryland
All eukaryotic cells undergo mitosis, in which newly replicated daughter DNA chromatids are equally segregated to the two opposite poles of the dividing cell. This process is wholly dependent on specialized chromatin containing the histone protein CENP-A. This protein has been reported to be over-expressed and mislocalized in cancer cells, partnering with inappropriate proteins. Thus, understanding the mechanical basis for CENP-A's unique function in the native and cancer state are fundamental questions in biology. Our project aims to use cutting edge cross-disciplinary tools such as all-atom simulations, using major supercomputing resources, coupled to high speed nano-imaging to visualize molecular changes in CENP-A in the native vs. cancer state.
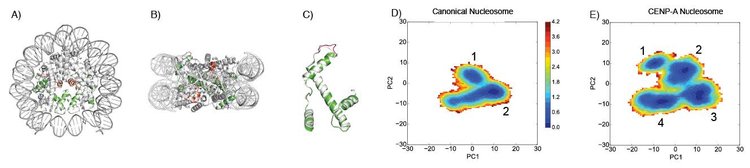
A-B) The CENP-A nucleosome crystal structure is shown in two orientations, where the CENP-A proteins are shown in green and the H2A acidic patches are shown in red. C) Structural alignment of H3 and CENP-A proteins show nearly identical geometry, despite very distinct biological functions of the corresponding nucleosomes. D-E) Free energy maps, calculated in Winogradoff et al (which was a result of a recent collaboration between Papoian and Dalal laboratories), indicate that a broader array of conformations are available to the CENP-A nucleosome, compared with the canonical, H3 nucleosome, providing the former with greater structural variability. In turn, this intrinsic plasticity may allow CENP-A nucleosomes to withstand eviction by being able to adapt and distort under biological forces such as mitotic tension.
A COMBINED COMPUTATIONAL AND EXPERIMENTAL APPROACH TOWARDS THE DESIGN OF A NOVEL NEXT-GENERATION MULTIVALENT HER3-SEQUESTRATION CANCER THERAPY
PRINCIPAL INVESTIGATORS:
Stanley Lipkowitz, National Cancer Institute
Steven Jay, Fischell Department of Bioengineering, University of Maryland
PH.D. STUDENT:
John Schardt, Bioengineering, University of Maryland
Dysregulation of signaling between protein ligands and their cell surface receptors is a primary cause of tumor growth and metastasis in many cancers. One example of this phenomenon, which is especially prevalent in breast cancer, is the overexpression of the receptor HER3 leading to increased dimerization of HER3 with another receptor, HER2, on the surface of cancer cells, resulting in pro-neoplastic effects. We have pioneered a strategy, HER3 sequestration, that utilizes engineered multivalent HER3 ligands to trap HER3 into configurations that prevent its interaction with HER2, thus inhibiting pro-neoplastic signaling. The goal of the proposed research is to employ computational protein design and cancer biology experimental approaches to better understand and enhance HER3 sequestration towards the development of a novel cancer therapeutic.
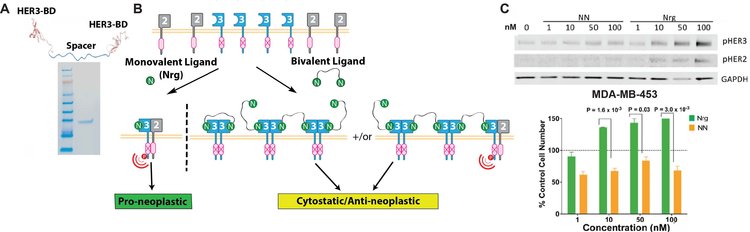
Overview of HER3 sequestration via multivalent ligands. (A) Schematic of a bivalent HER3 ligand identifying the key components: HER3-binding domains (HER3-BD) separated by a protease resistant spacer region. In this example, a bivalent protein consisting of 2 neuregulin-1β (Nrg) domains, which bind HER3 with high affinity, was purified to >98% as indicated by SDS-PAGE. (B) Multivalent HER3 ligands can be used to draw HER3 into non-productive HER3-HER3 homotypic interactions, contrary to HER3-HER2 interactions, which are proneoplastic and are prevalently stimulated in cancer by native, monovalent ligands such as Nrg. (C) In MDA-MB-453 breast cancer cells, monovalent Nrg stimulates phosphorylation of HER3 and HER2 in a dose-dependent manner leading to increased cell growth. Bivalent Nrg (NN) does not induce HER3 or HER2 phosphorylation and leads to decreased cell growth compared to control media conditions.